All Categories
Featured
Table of Contents
Buy Can Cannabis Help Patients With Parkinson's Disease? in TEXAS - limited period only
Pharmacokinetic studies are typically conducted in healthy controls. Persons with CNCP often have multiple physical comorbidities and are prescribed multiple medications. This study was conducted in a “real world” cohort which necessitated decisions regarding limitations on prior and concomitant treatments and/or drug interactions. THC induces CYP1A2 activity and reduces serum concentrations of drugs metabolized by CYP1A2, e.
anti-psychotic medications such as clozapine, olanzapine, haloperidol, and chlorpromazine [20]. Thus persons with psychosis were excluded from this study. Serum concentrations of duloxetine and naproxen may be reduced with concomitant use of THC: persons on these drugs were eligible for recruitment but were required to be monitored for drug interactions.
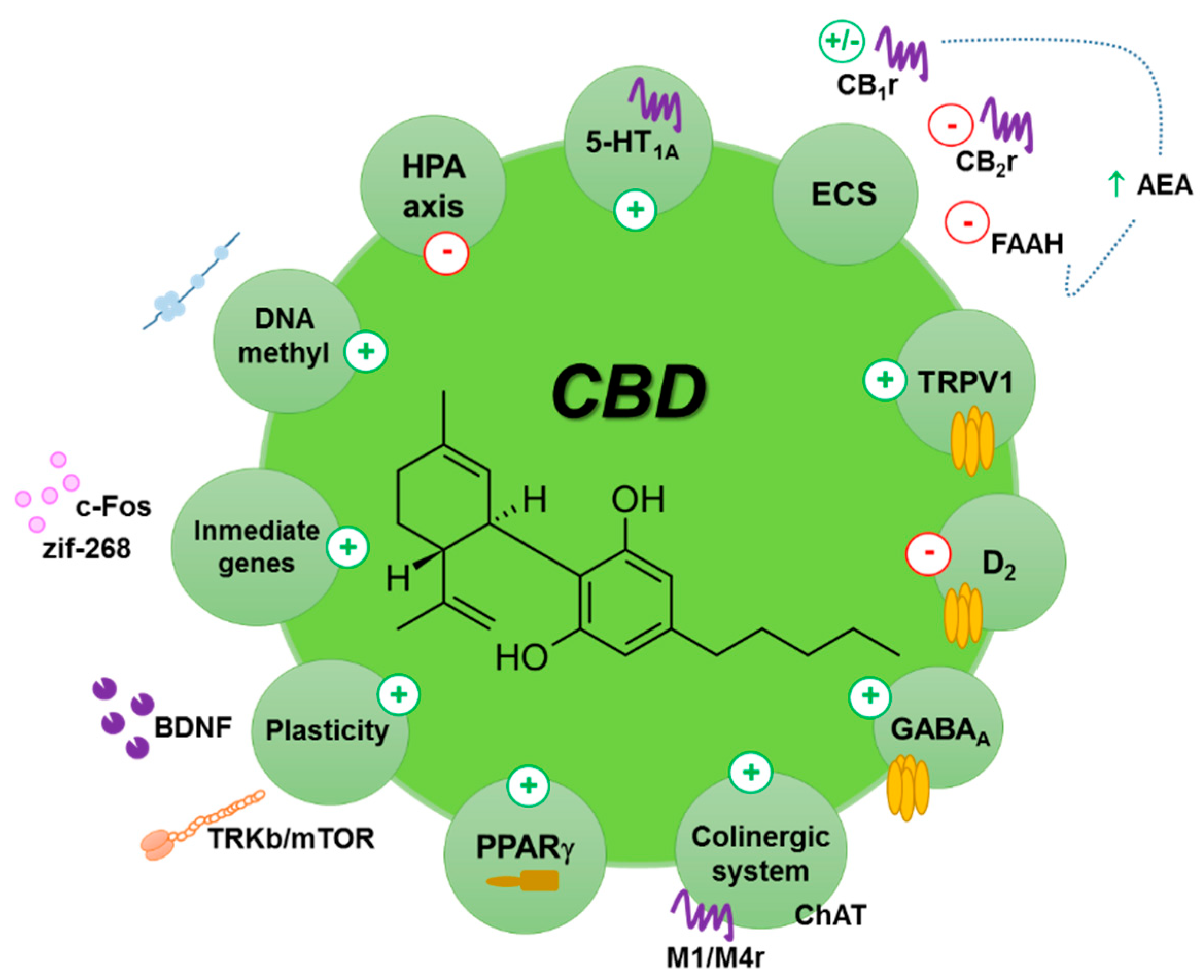

Tramadol metabolism is primarily mediated through CYP2D6, CYP3A4, and CYP2B6 [21]. Carbamazepine is a potent inducer of CYP3A4, and CBD may inhibit its metabolism resulting in increased serum concentrations. Serum concentrations of topiramate have also been observed to rise with increasing CBD dose but changes have been reported to be within the accepted therapeutic range [22].
Current use of valproate was an exclusion for the current study as this combination may elevate aspartate transaminase (AST) and/or alanine aminotransferase (ALT) concentrations [23]. Benzodiazepines were excluded because of potential excessive sedation in combination with the investigational product (ZTL-103). No new medication other than the investigational product, opioid medications for CNCP, and contraceptives was permitted to be commenced after initiation on to the trial.
Buy Cannabinoids In Late Life Parkinson's Disease And Dementia in LA - limited time
The pharmacokinetics were investigated on day 1 after a single dose of 2. 5 mg THC/2. 5 mg CBD after fasting for 12 h, day 8 after a single dose of 2. 5 mg THC/2. 5 mg CBD following a high fat meal, day 15 after a single dose of 5 mg THC/5 mg CBD after twice daily 2.
5 mg CBD for 7 days, day 22 after a single dose of 7. 5 mg THC/7. 5 mg CBD after twice daily 5 mg THC/5 mg CBD for 7 days, and day 29 after a single dose of 12. 5 mg THC/12. 5 mg CBD after twice daily 7.
5 mg CBD for 7 days. Participants returned for final assessments on day 36, after a 7-day washout period; however, no bloods were taken on this day as it was not anticipated that there would be any THC or CBD 1 week after a single dose of 12. 5 mg THC/12.
As an indication of this a single dose of 12. 5 mg THC/12. 5 mg CBD was given the previous evening at 4 pm prior to the day 29 pharmacokinetic (PK) analysis. The concentrations of THC and CBD in the predose sample approximately 20 h later were generally around 0.
Cannabidiol ( Drugbank - Ddrare now in NYC


Thus it would be highly unlikely that after a single dose of 12. 5 mg THC/12. 5 mg CBD there would be detectable THC and CBD 148 h later. To explore secondary objectives, participants completed the Brief Pain Inventory (BPI) [24] which has two subscales, namely pain severity and pain interference (i.
how pain impacts function and daily life), and recorded opioids, other analgesics, and any other medications, daily. At each visit to the study site (days 1, 8, 15, 22, 29, 36) participants completed the Depression, Anxiety, and Stress Scale (DASS-21) [25], Insomnia Severity Index (ISI) [26], and underwent a physical examination by a clinician.
All participants completed a daily sleep diary. Three variables were recorded: Sleep onset latency (SOL)—the participant’s perceived ease of falling asleep (Likert scale); Total sleep time (TST)—“Last night I slept a total of x hours”; Sleep quality—“When I woke up for the day I felt: refreshed, somewhat refreshed or fatigued”.
Bloods were refrigerated at 4 °C or below, and centrifuged at 2000×g for 10 min within 12 h after sampling. Immediately after centrifugation, plasma was stored in two labelled polypropylene tubes and stored at − 20 °C or below for plasma concentration analysis. All plasma concentration analyses were performed after all participants had completed the final visit.
September 28, 2021 - Cu Anschutz now in NC - limited time only
On days 1, 15, 22, and 29 participants were offered decaffeinated beverages and digestive biscuits from 15 min post-dosing. They were provided with lunch at 4 h post-dose and a light meal late afternoon. On day 8, participants were provided with a high fat breakfast prior to dosing. The breakfast was consistent with the US Food and Drug Administration guidelines for fat and calorific content [27], providing an energy value of approximately 3760 k, J (900 kcal) and containing approximately 60 g of fat.
Table of Contents
Latest Posts
Buy The 13 Best Heart Rate Monitors Of 2023 - Verywell Fit in LA
Garmin Hrm-pro Review: An Accurate Heart Rate Monitor now in TEXAS - limited time only
Heart Rate Monitors - Walmart.com now available in LA - limited time
More
Latest Posts
Buy The 13 Best Heart Rate Monitors Of 2023 - Verywell Fit in LA
Garmin Hrm-pro Review: An Accurate Heart Rate Monitor now in TEXAS - limited time only
Heart Rate Monitors - Walmart.com now available in LA - limited time